Since founded, our group has built a relatively complete facility and fine research environment. We have published more than 50 papers, 10 granted papers, and 10 book chapters. In a free atmosphere, our group give full play to every individual’s our talent and cultivate his or her research capacity and perspective.
1. Synthesis and characterization of high-strength hydrogel
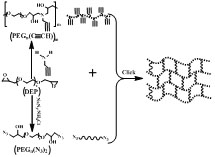

Amine-epoxy click polymerization (AECP) is used to prepare novel PEG-based hydrogels with well-defined networks, improved mechanical properties and increased compatibility. To achieve this goal, α, ω-diepoxy PEG was a starting material for synthesizing other PEG derivatives.Alkyne-functionalized linear PEG derivatives with controllable alkyne loadings and variable molecular weight have been prepared by propargylamine and α, ω-diepoxy PEG of different molecular weights under controlled conditions. Then, α, ω-diazide/hydroxyl PEG was synthesized via the terminal epoxy groups of α, ω-diepoxy PEG through a series of reactions to more active azide groups. Finally, PEG-based hydrogels can be obtained through the formation of covalent networks using CuAAC of different molecular weight linear alkyne- functionalized linear PEG derivatives and α, ω-diazide/hydroxyl PEG. We present and discuss the results that characterize and compare PEG-based hydrogels in swelling ratio, mechanical properties, thermal properties,degradation and cytocompatibility in this report for possible biomedical applications.
2. Well-defined Amphiphilic Polymer Conetwork from Precise Control of the End-Functional Groups of Linear RAFT Polymers
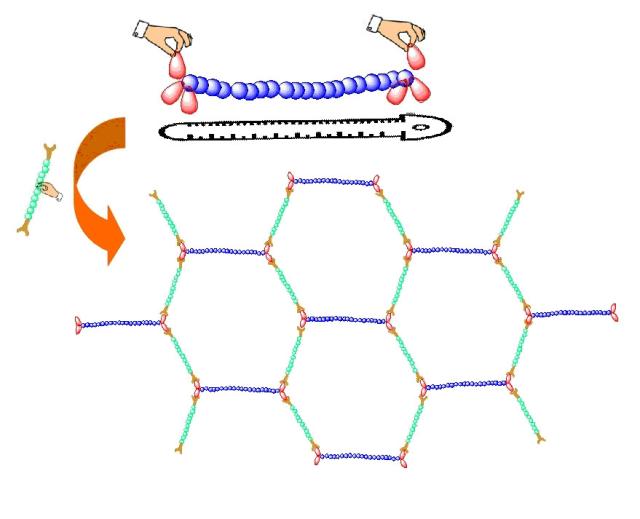
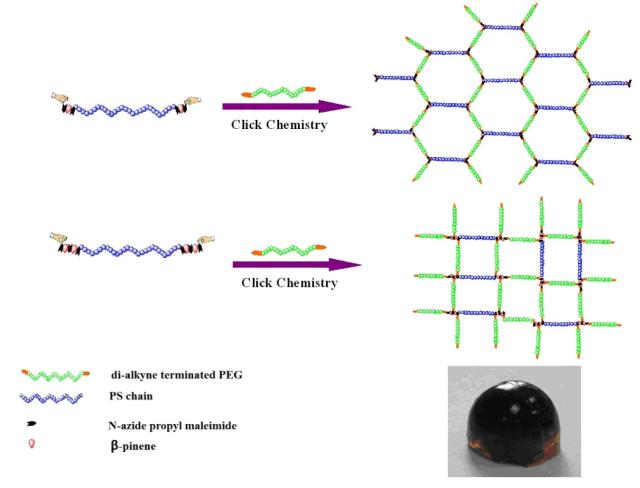
Linear polystyrene (PS) with well-defined molecular structure and accurate number of bromo groups on both ends were synthesized via multiple-step alternative RAFT polymerization of N-bromopropyl maleimide and β-pinene monomers. The bromo end groups were transformed into the azido moieties via nucleophilic substitution. The reaction of as-synthesized linear PS having named number of azide groups on ends ((N3)x-PS-(N3)x) with mono- and dialkynyl-terminated PEG (dA-PEG) via copper (I)-catalyzed azide-alkyne cycloaddition (CuAAC) leads to the formation of the well-defined PS-PEG amphiphilic copolymers and polymeric conetworks (APCNs). The as-prepared APCNs exhibit unique ordered separated hydrophilic and hydrophobic phases, and a variable swelling capacity both in polar and non-polar solvents.
3. Precise control of structure of polymer network
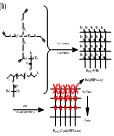
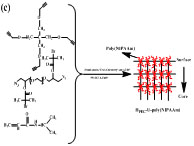
A series of well-defined hydrogels with precisely controlled molecular structures including hydrogels with polymer brushes on surface, hydrogels with gradient distributed polymer brushes from surface to inside and hydrogels with polymer brushes uniformly distributed in whole polymer network were prepared via combining CuAAC with ATRP in the presence of modified PEG2000 serial compounds, 2-bromoisobutyryl bromide (BiBB), Tetrakis(2-propynyloxymethyl)methane (TPOM), copper(I)-bromide (CuBr), pentamethyldiethylenetriamine (PMDETA) and N-isopropylacrylamide (NiPAm) using bromide from 2-bromoisobutyryl bromide as initiator, copper(I)-bromide as catalyst, and PMDETA as ligand. The hydrogels with well-defined molecular structures exhibit a fast response to the change of the surround temperature, due to the thermosensitivity of the polymer brushes. These hydrogels could be found potentially applications in biosensing, intelligent diagnosis, proximity warning and so on.
4. Controlled Click-chemistry and its application in controlling hydrogel structure